
Nonmetal atoms gain electrons to form anions with a negative charge equal to the A group number minus eight. In the long form of the periodic table, the valence shell electronic configuration of 5s☥p4 corresponds to the element present in: Group 16 and period 6 Group 17 and period 6 Group 16 and period 5 Group 17 and period 5 Answer: Group 16 and period 5. Ion Electron Configurations Metal atoms lose electrons to form cations with a positive charge equal to the group number. The number of elements in the 5th period of the periodic table is. Half-filled d subshells are favored if possible (Hund’s Rule) 3dĮxample 5 What family of elements is characterized by having an ns2np3 valence-electron configuration? Group 5A X has ns2 np1, in ground state, its excited state will be ns1 np2 i.e 2 unpaired electron, now Y has 3 unpaired electron already in ground state.
NS2 NP3 VALENCE SHELL PLUS
Valence electrons – electrons held in outer shell Core electrons – electrons held in filled inner shellsĮlectron Configurations of Transition Elements ĭ subshell being filled valence electrons - s and p electrons in outermost shell plus electrons in incompletely filled (n - 1)d subshell (n - 1)d orbitals are filled after ns orbitals and before filling np orbitals Let's use the second period for this for a for one veiled Solectron, we're gonna use the element lithium and its electron configuration would be one s two, uh, to s one foreign core notation.

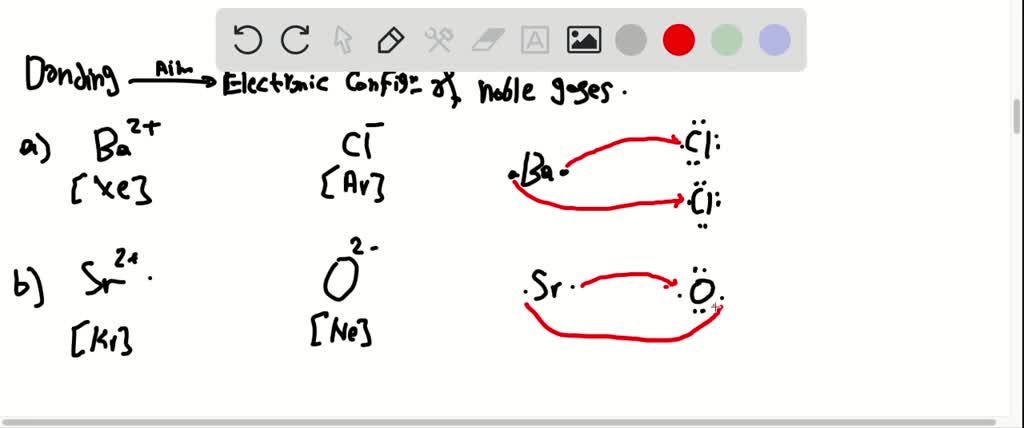
Valence Electrons Chemically similar behavior occurs among elements within a group in the periodic table. through to the elements were asked to given Adam and it's complete electron configuration that would be expected to have communicated number of valence electrons. Pnictogens have multiple allotropic forms resulting from their ns2 np3 valence electronic configuration, making them the only elemental materials to crystallize in layered van der Waals (vdW) and quasi-vdW structures throughout the group. go back and pair electrons with 4, 5 and 6 Hund’s Rule – electrons pair only after each orbital in a subshell is occupied by a single electron C: 1s 2 2s 2 2p2 1sĮlectron Configurations and the Periodic Table Pnictogens Allotropy and Phase Transformation during van der Waals Growth.Preceeding the noble gases are two chemically important groups of non-metals such as halogens (Group 17) and the chalco gens (Group 16). This configuration is highly stable and hence noble gases exhibit very low chemical reactivity. half-fill each orbital with first 3 electrons At the end of each period is a noble gas element with a closed valence shell ns2 np6 configuration.Win over the concepts of Chemistry and get a step ahead with the preparations for General Science with Testbook. for atoms in ground state – electrons occupy energy shells, subshells and orbitals that give the lowest energy for the atom Įlectron Configurations for orbitals with same energy (degenerate) such as the three 2p orbitals The electron configuration ending in ns2 np3 belongs to group VA (the superscripts total to '5' indicates there are 5 valence electrons for elements belonging to Group VA).Elements in other groups vary in their reactivity but are generally less reactive than elements in groups \(1,\,2,\,16,\) or \(17.\) Valence Electrons and Electricity 1.Electron Configuration – complete description of orbitals occupied by all the electrons in an atom

NS2 NP3 VALENCE SHELL FULL
These elements already have a full outer energy level, so they are very stable. 31) Consider the general valence electron configuration of ns2 and the. Atoms of group 18 elements have eight outer-shell electrons (or two in the case of helium). 7) The electron configuration of the atom with the largest atomic radius is. These subatomic particles carry a negative charge of \(1.602 \times \right),\) it readily accepts the electron given by the sodium atom. The elements X, Y, Z and J have the indicated electron configurations. Valence Electrons: Concepts to UnderstandĮlectrons are tiny subatomic particles that revolve around the nucleus in energy shells called orbits.

